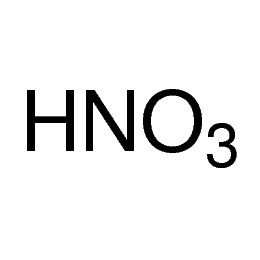
Nitric acid 70%, purified by redistillation, ≥99.999% trace metals basis
Nitric acid is generally used as a primary reagent for the nitration of organic molecules. It is also used as a strong oxidizing agent in organic synthesis to perform the oxidative transformations.[3] [4][5]
Nitric acid used as a reagent in:
• The nitration of aromatic compounds, aromatic heterocycles, alkenes, active methylene carbons, etc.[3][6]
• Selective oxidation of organosulfur compounds to corresponding sulfoxides using a solid acid catalyst.[7]
•
The oxidation of alcohols, aldehydes, esters, and heteroatoms.[3]
• The aromatization and dehydrogenation reactions.[3]
475 mL in glass bottle